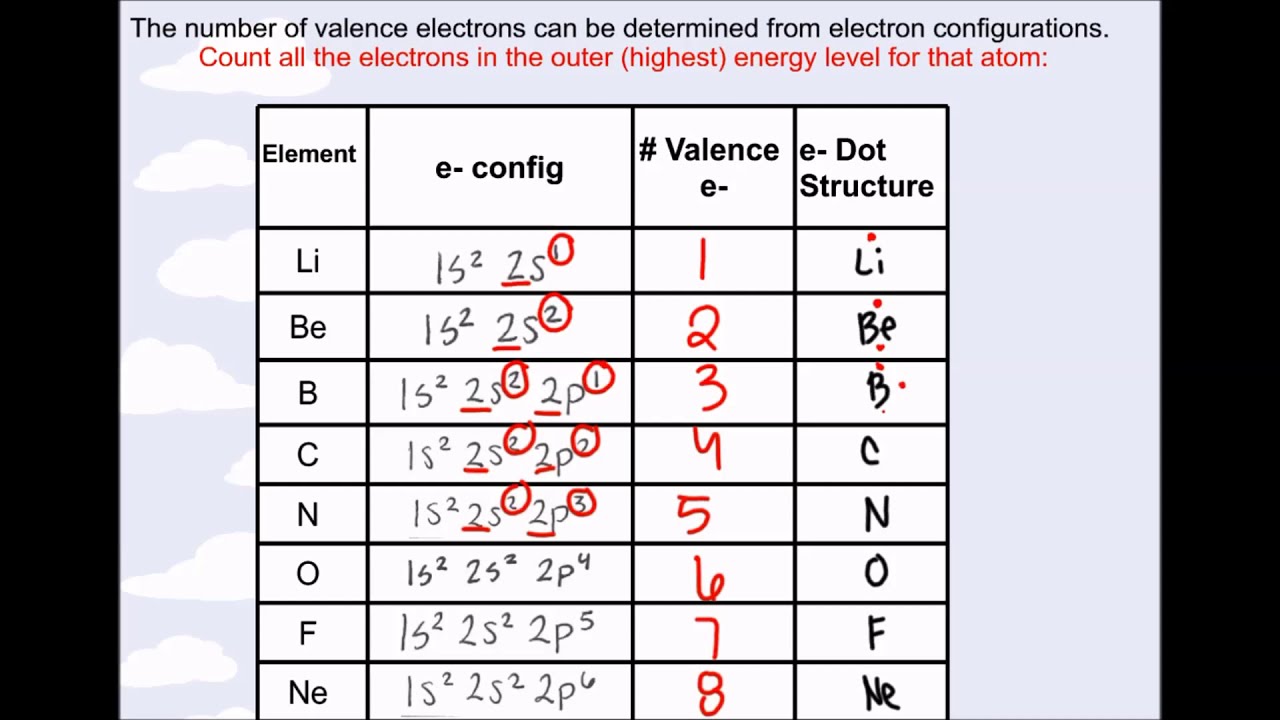
With the help of group numbers, we can easily calculate the number of valence electrons of an element.Valence electrons can be determined by checking the vertical column of the periodic table.The structure of these elements could be much better. The electrons of these groups can be determined easily, but they are difficult to predict. It can be found in all elements, even if they are from Group 3 to Group 12. Energy level 2 has 2 subshells (orbitals) s and p each holding 2 and 4 electrons respectively.Valence electrons can easily be determined with the help of the periodic table.Energy level 1 has 1 subshell s which can contain 2 electrons.So to just clarify again, In oxygen we have 2 energy levels - 1 and 2. You need to note that s and p are orbitals in the same Energy Level (2) so we need to take addition of all electrons available in those orbitals. Most of the students get confused with orbital and energy level. So total electrons in the outermost shell is 6. 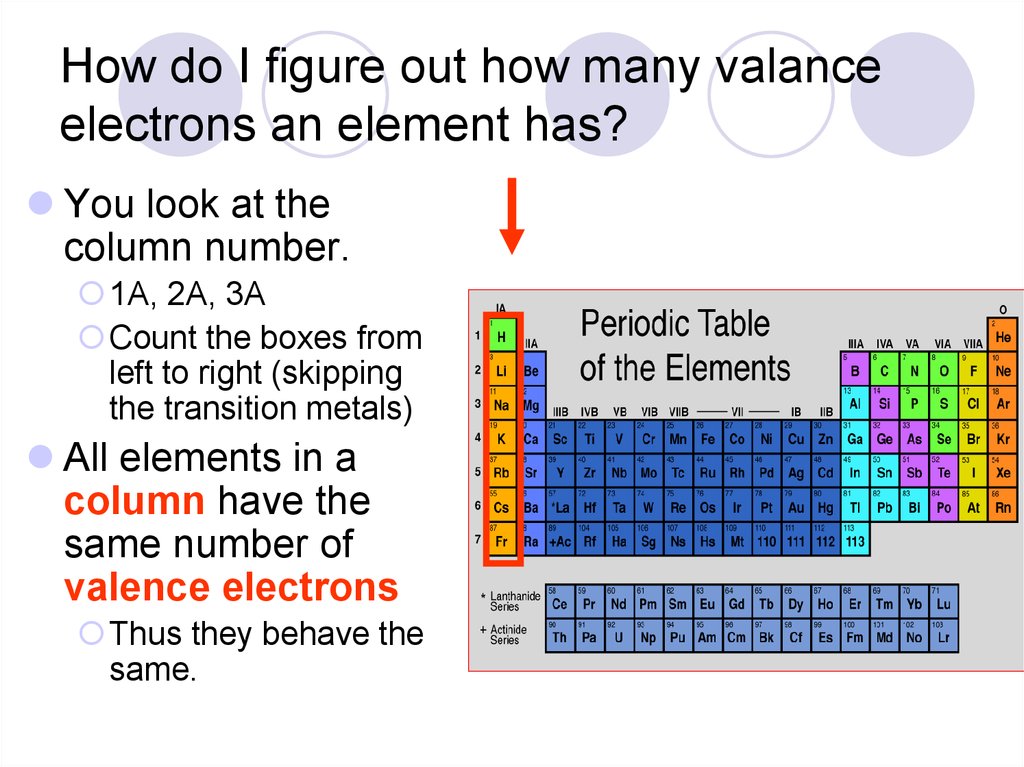
So Oxygen (O) has 6 valence electrons because outermost shell or energy level is 2 and in that energy level we have 2 and 4 electrons in s and p orbital respectively. ExampleĮach oxygen atom has the electron configuration 1s² 2s² 2p⁴ Understanding the valence electrons of an atom is crucial for predicting its chemical reactivity and bonding behavior in chemical reactions. Hydrogen has 1 valence electron, and helium has 2 valence electrons.īy following these steps and referring to the periodic table, you can determine the number of valence electrons for most elements. Some elements, like hydrogen and helium, do not fit the typical valence electron pattern because they have only one energy level. You may need to refer to a chemistry resource or table to determine the valence electrons for transition metals. Their electron configuration may involve filling the d orbitals, and the number of valence electrons can vary depending on the specific element and its oxidation state. Transition metals, found in the middle of the periodic table, have variable valence electrons. Group 18 elements (noble gases) have 8 valence electrons, except for helium, which has 2 valence electrons.Group 17 elements (halogens) have 7 valence electrons.Group 16 elements have 6 valence electrons.Group 15 elements have 5 valence electrons.Group 14 elements have 4 valence electrons.Group 13 elements have 3 valence electrons.Group 2 elements (alkaline earth metals) have 2 valence electrons.Group 1 elements (alkali metals) have 1 valence electron.The number of electrons in the valence shell corresponds to the group number of the element on the periodic table.This level is often referred to as the "valence shell." To find the valence electrons, focus on the electrons in the outermost energy level.Count the Electrons in the Outermost Shell The electron configuration tells you how the electrons are distributed in different energy levels or electron shells.  
You can find the electron configuration of an element using the periodic table or reference materials. The atomic number is typically found on the periodic table and is unique to each element. Look up the atomic number of the element you are interested in. Here's how you can find the valence electrons of any atom: Determine the Atomic Number Valence electrons are the electrons in the outermost energy level or shell of an atom. Home iit-jee how to find the valence electrons of any atom how to find the valence electrons of any atomįinding the valence electrons of an atom is essential for understanding its chemical behavior and its potential to form bonds with other atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
